What is the scientific rationale and what does the evidence show?
by Adam Denny, PhD
In recent years, scientists around the world have begun to investigate the role of oxidative stress in FSHD. A 2018 study reported that muscle biopsies from people with FSHD had greater levels of oxidative stress markers when compared to healthy control muscle (Dias Wilson et al., 2018). In another study, a stem cell-based model indicated that high oxidative stress itself can lead to increased DUX4 expression (Sasaki-Honda et al., 2018).
These findings indicate that DUX4 can increase oxidative stress, and that oxidative stress itself can increase DUX4 expression, proposing a damaging feedback loop, although this hypothesis needs further investigation.
What is clear in FSHD is the imbalance in reactive species and antioxidant defenses, the two key aspects underpinning oxidative stress.
What is oxidative stress?
Oxygen is essential to life, but it’s a double-edged sword. Certain oxygen-containing molecules are chemically very reactive and, if allowed to run amok in our bodies, can damage DNA, proteins, fats, and other components. To combat these “reactive species,” biology has evolved molecules called antioxidants that neutralize or repair the damage.
Oxidative stress results when the production of reactive species outpaces the body’s antioxidant defenses. This imbalance can result in longterm damage. Increases in oxidative stress are beneficial in some circumstances, for example, to destroy cancer cells, but in disease, oxidative stress can be harmful.
The role of oxidative stress in FSHD
Most studies of FSHD and oxidative stress are conducted in laboratory models, so we have to be cautious about extrapolating these findings to humans.
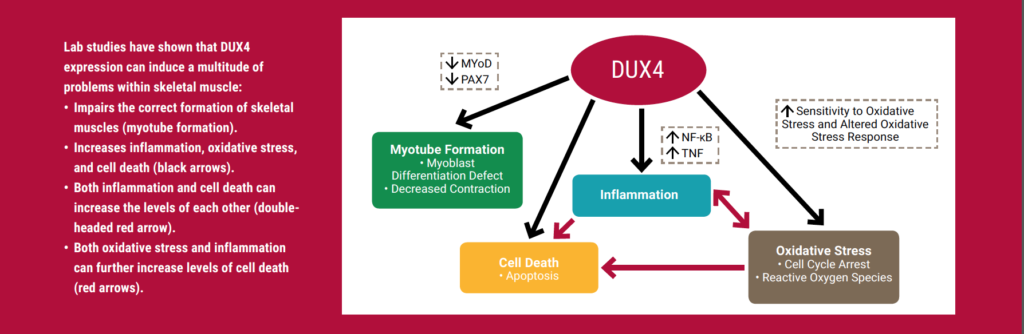
As depicted in the figure (above), lab studies have shown that DUX4 expression can induce a multitude of problems within skeletal muscle. These include impaired formation of muscle cells and increased inflammation, oxidative stress, and cell death.
Given these lab findings, individuals with FSHD may well wonder if they should take steps to reduce the potential harm from oxidative stress.
About antioxidants
To combat oxidative stress, we have antioxidants—substances that can delay, prevent, or remove oxidative damage.
There are two categories of natural antioxidants: enzymatic and nonenzymatic antioxidants. Enzymatic antioxidants are produced by our bodies and work through breaking down and protecting against the reactive species. Non-enzymatic antioxidants are a much larger class of antioxidants and work through disrupting the chemical reactions caused by reactive species.
Some natural non-enzymatic antioxidants are made within our bodies, such as vitamin A and coenzyme Q10. When our bodies don’t make sufficient amounts, we can take them in pill form. Others stem from external sources, such as vitamins C, E, and K, along with zinc and selenium.
Antioxidants from external sources are important to factor into a person’s diet. While these antioxidants are also available as pill supplements, many people prefer to obtain these antioxidants from a balanced diet.
Antioxidant trials
While antioxidants have shown promise throughout the years in laboratory studies, these results have been hard to replicate in clinical trials.
 One clinical trial investigated the effect of combined dietary supplementation of vitamins C and E, and minerals zinc and selenium in individuals with FSHD (Passerieux et al., 2015). The researchers found that supplementation did not improve the two-minute walk test, but it did improve muscle function: individuals could contract their thigh muscles (quadriceps) harder and for longer than they could before the supplementation.
One clinical trial investigated the effect of combined dietary supplementation of vitamins C and E, and minerals zinc and selenium in individuals with FSHD (Passerieux et al., 2015). The researchers found that supplementation did not improve the two-minute walk test, but it did improve muscle function: individuals could contract their thigh muscles (quadriceps) harder and for longer than they could before the supplementation.
The participants who received supplementation also saw decreases in some of their oxidative stress markers and increases in certain markers of their antioxidant defense system.
In another small clinical trial, a Dutch group (Van der Kooi et al., 2016) investigated the supplementation of folic acid and methionine in people with FSHD. Both supplements have antioxidant properties, and folic acid has previously been shown to boost DNA methylation, but this study found neither folic acid nor methionine had an effect on DNA methylation levels.
Other ways to protect against oxidative stress
 Exercise is a very effective way to increase our antioxidant defenses. However, in order to obtain this benefit, one has to first use exercise to increase levels of oxidative stress. The body then responds by boosting its antioxidant defenses.
Exercise is a very effective way to increase our antioxidant defenses. However, in order to obtain this benefit, one has to first use exercise to increase levels of oxidative stress. The body then responds by boosting its antioxidant defenses.
While the oxidative stress produced in exercise is vital, the exact effect this may have on FSHD is not fully understood. The type, intensity, and duration of the exercise all may influence the outcome.
Bankolé and colleagues (Bankolé et al., 2016) have shown that a combined strength and interval cycling exercise-training program significantly improved fitness and skeletal muscle function without negatively impacting muscle damage. While this study did not investigate the antioxidant levels of participants, it does highlight that certain types of exercise can be beneficial for people with FSHD regardless of the increases in oxidative stress induced by exercise.
In conclusion…
Research to date has shown some positive results, but important questions remain. First, oxidative stress has a strong link with FSHD cells in the test tube, but the relevance of oxidative stress in FSHD patients is unknown.
Also, while antioxidant therapies have shown modest benefits, these results came from relatively small clinical trials. We need to conduct larger trials to understand if antioxidant therapies can protect muscles in people with FSHD.
Editor ’s note: Adam Denny earned his PhD in the Department of Physiology at the University of Otago in New Zealand.



Thank you for this complete article on that interesting subject! And thank you for highlighting french research 😉
I wonder whether a balanced diet is enough to counteract oxidative stress or if the doses needed are too important and necessitate artificial supplementation.
Sylvie (from patient FSH group in AFM-telethon France)
I would really suggest you should talk to Prof dalila.laoudj-chenivesse@inserm.fr
She is been the very first scientist to investigate anti-oxidation and FSHD, and she has made a positive impact on the life of many FSH patients including my son.
Best regards.
What dosage of the various mentioned vitamins would be useful to take daily for an adult man of 70 years of age suffering from FSHD
Would
really appreciate some advice